*This post is for Thursday, April 28th.*
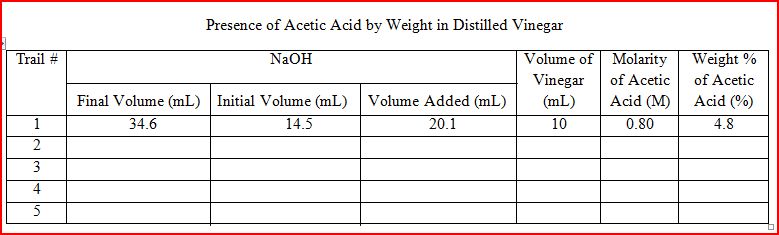
In today's experiment, we tested the weight percentage of acetic acid in vinegar by reaching the point of equilibrium between CH3COOH and NaOH. Unfortunately, we were only able to complete one successful trial, but we intend to add to our data soon.
In today's experiment, we tested the weight percentage of acetic acid in vinegar by reaching the point of equilibrium between CH3COOH and NaOH. Unfortunately, we were only able to complete one successful trial, but we intend to add to our data soon.
| Calculations Molarity of Acetic Acid: M = 0.40 (20.1) / 10.0 mL M = 0.80 M | Mass of Acetic Acid/Liter: m = 0.80 x 60.06 m = 48 grams | Weight Percentage of Acetic Acid: w% = (48/1000) x 100 w% = 4.8% |
-Gretchen Koomjohn

 RSS Feed
RSS Feed
